Chemistry - Unit 1 - OCR
The Atom:
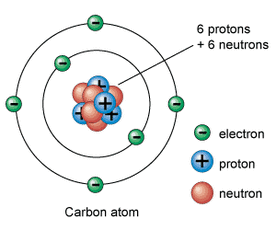
Atoms are made up of Protons, Neutrons and Electrons.
Electrons: have a -1 charge. Electrons move around the nucleus in orbitals. The orbitals take up most of the volume of the atom.
Nucleus: this is where most of the mass of the atom is concentrated. The volume of the nucleus is small in comparison to that whole atom. The nucleus is where the protons and neutrons are found.
The relative charges and relative masses of these subatomic particles is shown below:
Protons: Relative mass: 1 Relative charge: +1
Neutrons: Relative mass: 1 Relative charge: 0
Electrons: Relative mass: 0.0005 Relative charge: -1
Electrons: have a -1 charge. Electrons move around the nucleus in orbitals. The orbitals take up most of the volume of the atom.
Nucleus: this is where most of the mass of the atom is concentrated. The volume of the nucleus is small in comparison to that whole atom. The nucleus is where the protons and neutrons are found.
The relative charges and relative masses of these subatomic particles is shown below:
Protons: Relative mass: 1 Relative charge: +1
Neutrons: Relative mass: 1 Relative charge: 0
Electrons: Relative mass: 0.0005 Relative charge: -1
Negative ions have more electrons than protons.
Positive ions have fewer electrons than protons.
Isotopes are Atoms of the same element with different numbers of Neutrons.
Positive ions have fewer electrons than protons.
Isotopes are Atoms of the same element with different numbers of Neutrons.
- The number and the arrangement of electrons decide the chemical properties that particular element has. Isotopes have the same configuration of electrons so they therefore have the same chemical properties.
- Isotopes of elements have slightly different physical properties e.g. variations in densities. This is because physical properties depend more on the mass of an atom.
Atomic models:
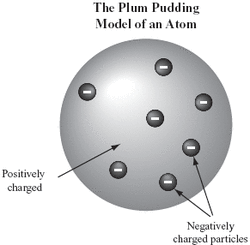
- Ancient Greeks thought that all matter was made from indivisible particles.
- At the beginning of the 19th century, John Dalton describe atoms as solid spheres, and said different types of sphere, made up different elements.
- As scientists started to perform more experienced, the currently accepted model began to emerge.
In 1897, JJ Thompson concluded that atoms weren't sold and indivisible:
- Measurements of charge and mass showed that atoms must contain smaller particles, that are negatively charged.
- The new "plum pudding model" was introduced - a positively charged sphere with positively charged
